|
|
|
|
|
|
Before we're done with this program we will have learned about electric charges and the forces acting on them. We will see how light can be analyzed to reveal the composition of distant stars and the internal structure of the atom. Then we will turn our attention to cathode rays and understand how they led to the discovery of the electron as a piece of an atom. After that we will look to the accidental discoveries of the mysterious and penetrating xrays,followed shortly by a likewise accidental discovery of radioactivity.
1.1.1. Describe the amber effect
1.1.2. Summarize the contributions of Gilbert and Franklin
1.1.3. State the law of conservation of charge
1.1.4. State the two basic properties of electric charges.
1.1.5. How do we know that there are only two kinds of charge?
1.1.6. Assuming that an object is charged, explain how you would determine whether the charge is positive or negative.
1.1.7. Describe the principle of operation of a simple battery (voltaic pile).
1.1.8. Volta's invention of the battery revolutionized the study of electricity. Why?
1.1.9. State Couloumb's Law of Electric Force.
1.1.10. Describe some of the similarities and differences between electric forces and the gravitational forces.
1.1.11. Why is it that we don't normally notice the strong electric forces between charges?
1.2.1. What is spectroscopy?
1.2.2. What is the difference between a continuous and a discrete spectrum?
1.2.3. What are Fraunhofer lines?
1.2.4. Compare the emission and absorption spectrum of a gas such as hydrogen.
1.3.1. What did Balmer discover about the hydrogen spectrum?
1.3.2. In the following formula, what does each of the terms represent?

This lesson could have been titled, "Pieces of Atoms". It is odd what comes flying out of atoms when you zap them with high voltage electricity.
The interest and research into electricity began in earnest with the invention of the battery in 1800. For the first time it was possible to produce and control a steady stream of electricity. We have already noted how this spurred the development of chemistry, especially in the discovery of new elements through electrolysis.
Now we add another line between chemistry and physics. We have seen how the concept of atoms combined with Newton's laws gave us the kinetic molecular theory of matter. This strengthened the physical science paradigm, and also provided new avenues for research into the nature of the atom.
It was the understanding of the electromagnetic nature of light which cemented the paradigm, and ultimately led to the quantum model of the atom. Several new discoveries at the end of the nineteenth century revealed a remarkable relationship between energy, light, electricity, and atoms.
Who would have guessed that electricity and light would become the key to understanding the structure of the atom.
1.4.1. properties of the atom were discovered which would provide the missing links
1.4.2. spectroscopy, electron, x-rays, radioactivity
1.5.1. led to understanding of the structure and nature of the atom and chemical elements
1.5.2. motion link still missing in explaining chemical bonding
1.5.3. chemical properties of the elements eventually understood in physical terms
2.1.1. Kinetic Theory and Atomic Theory together give us a picture of matter in motion, freedom of motion determines state
2.1.1.1. kinetic theory deals with physical properties of atoms and molecules
2.1.1.2. atomic theory deals with chemical properties of atoms and molecules
2.1.1.3. both allow for attractive forces between the atoms and molecules
2.1.1.4. neither tells us about the atoms themselves or the forces acting between them
2.1.1.5. first suggestion of electrical nature was by Berzelius in 18172.1.1.5.1. great Swedish analytical chemist
2.1.1.6. by the beginning of 20th century it was clear that the electrical nature of matter required a new concept
2.1.1.6.1. charge can not be defined in terms of more elementary concepts, like force, energy, power
2.1.1.6.1. must be intuitively derived as were mass, length, and time
2.1.1.6.3. by experiment on the properties of charges: behavior, magnitude, laws2.1.2. Early knowledge of what we now know to be electrical phenomena was of four types
2.1.2.1. the amber phenomenon
2.1.2.2. St Elmo's fire
2.1.2.3. Lightning
2.1.2.4. torpedo fish and electric eel "stings"2.1.3. They saw no connection
2.1.4. Little new added from Thales to Gilbert
2.1.5. Gilbert thought it was like magnetism, couldn't distinguish the effects
2.1.6. Stephen Gray (early 18th) conducted a charge over a tenth of a mile through wet twine and showed that the power of attraction could be transferred from on metallic object to another
2.1.7. Charles Dufay in France demonstrated that there were two and only two kinds of electricity: vitreous (glass rubbed with silk) and resinous (amber rubbed with wool). Each attracts the other and is self repulsive2.1.7.1. two imponderable fluids, each transferable by rubbing
2.1.7.2. amber/fur: resinous fluid transferred to amber leaving fur with excess of vitreous fluid
2.1.7.3. a few years later it was shown that resinous kind could be produced by rubbing clean glass with flannel2.1.8. Benjamin Franklin rejected two fluids
2.1.8.1. normal substance contained a normal amount of single fluid
2.1.8.2. more or less would produce positive or negative charge
2.1.8.3.2.1.9. Either theory can explain the electrostatic properties known at this time
2.1.9.1. Franklin's was more parsimonious
2.1.9.2. still use "juice" and "positive/negative" for electrical terms2.1.10. Benjamin Franklin was asked "what good is it". His reply, "of what good is a newborn babe"
2.1.10.1. Important Phenomenon
2.1.10.1.1. modern society depends on it
2.1.10.1.2. provides the missing link between physics and chemistry
2.1.10.1.3. helps us to gain a clearer picture of the structure of the atom
2.1.10.1.4. and the nature of chemical properties and reactions through the concept of chemical bonding2.1.10.2. no one could have imagined the uses we have put it to
2.1.10.3. methods of science applied to study in late 1700s2.1.10.3.1. rapid growth of understanding
2.1.10.4. later connections with chemistry, heat, energy, magnetism, light
2.2.1. objects can be "electrified" when rubbed with other objects
2.2.1.1. once electrified can attract other objects
2.2.1.2. "static cling" in laundry2.2.2. electros = amber
2.2.3. discovery noted around 600 B.C.
2.2.4. attracted pieces of straw, leaves, etc.
2.2.5. not much use for anything2.2.5.1. not produced in large amounts
2.2.5.2. quickly lost in damp air2.2.6. The video program demonstrates some causes and effects of static electricity.
2.3.1. William Gilbert (1600) De Magnete
2.3.1.1. described everything known about electricity and magnetism
2.3.1.2. described his experiments
2.3.1.3. conclusions and speculations
2.3.1.4. demonstrated amber is not the only electrifiable material2.3.1.4.1. attracted various types of material, not just straw and grass
2.3.1.5. two different kinds of materials
2.3.1.5.1. glass, porcelain
2.3.1.5.2. amber, silk2.3.2. Benjamin Franklin (1706-1790)
2.3.2.1. proposed two kinds of charges, positive and negative
2.3.2.1.1. glass acquires positive charge when rubbed with silk
2.3.2.1.2. rubber acquires negative charge when rubbed with fur2.3.2.2. law of electric charges: like charges attract, unlike attract
2.3.2.3. showed lightning was a form of electricity2.3.2.3.1. flew kite in thunderstorm
2.3.2.3.2. several others killed trying to repeat2.3.3. charge conservation
2.3.3.1. another conservation law
2.3.3.2. charge lost by one object (substance) equals charge gained by another
2.3.3.3. charge cannot be created or destroyed
2.3.3.4. total amount of charge in universe is constant2.3.4. electrostatic series
2.3.4.1. when rubbed together each material becomes more positive than the one below it
glass
mica
wool
quartz
fur
silk
felt
cotton
wood
cork
amber
graphite
rubber
sulfur
2.4.1. some substances transmit electric charges easily, others do not
2.4.2. wide range of conductivities, punctuated rather than continuous2.4.2.1. materials are generally either conductors or insulators
2.4.2.1.1. metals conduct, nonmetals do not
2.4.2.2. semiconductors are small group which falls between the two
2.4.2.2.1. silicon and germanium are most common examples
2.4.2.2.2. conductivity increases when illuminated or when temperature is raised
2.5.1. Charles de Coulomb (1736-1806)
2.5.1.1. used a torsion balance to determine magnitude of force between charges
2.5.1.2. served as model for Cavendish's gravitational torsion balance later2.5.2. Coulomb's Law

2.5.2.1. inverse square relationship
2.5.2.1.1. force between charges is proportional to charges, inversely proportional to square of distance
2.5.2.2. same mathematical form as Newton's gravitational law
2.5.2.2.1. involves charge instead of mass
2.5.2.2.2. gravity only attracts, electric force may also repel
2.5.2.2.3. electrical forces are much stronger than gravity2.5.2.2.3.1. about 1x1040 as much for two protons
2.5.2.2.4. normally not noticed because matter generally has equal amounts of positive and negative charges
2.5.2.2.4.1. opposite charges "cancel" each other out
2.6.1. Luigi Galvani (1737-1798) studied actions of nerves and muscles
2.6.1.1. accidental discovery while "drying" frogs
2.6.1.1.1. touching two different kinds of metal made legs kick
2.6.1.2. assumed a special kind of "animal electricity"
2.6.1.2.1. animal tissue must touch the two metals to produce it
2.6.2. Alessandro Volta (1745-1827) repeated Galvani's experiments
2.6.2.1. Galvani sent Volta his results and thoughts
2.6.2.2. constructed sensitive electroscope
2.6.2.3. produced electric current with two different metals and salt water2.6.2.3.1. not special "animal" electricity, but inorganic phenomenon
2.6.2.4. invented "voltaic pile" (first battery)
2.6.2.4.1. zinc/silver in contact with salt water
The First Battery: Volta's Electric Cell
2.6.2.5. major observations
2.6.2.5.1. stack of 20 or so "cells" produced vigorous deflection of the electroscope
2.6.2.5.2. the electroscope is a device for detecting unbalanced charge
| The electroscope. A metal rod is fitted into a stoppered flask. I consists of two thin pieces of gold foil which swing freely on a hinge. When an electric charge is put on the metal ball they flow into the gold foil. Because they are charged with like charges they repel one another. The distance of the swing, or the angle of separation is proportional to the charge. |  |
2.6.2.5.3. wire connected across opposite plates became hot
2.6.2.5.3.1. plates became hot too
2.6.2.6. effects
2.6.2.6.1. electrolysis of water: water is not an element
2.6.2.6.2. electricity and heat: Joule's experimentJoules's Law of Heating. Electric power is proportional to the square of the current.
2.6.2.6.3. electricity and magnetism: Faraday's motor and generator
2.6.2.6.4. electricity and chemistry: electroplating (also Faraday)
2.6.2.6.5. animal and metallic electricity are the same (also Faraday)2.6.2.6.5.1. modern examples include heart beats, brain waves, nerve impulses
2.6.2.6.6. dry cell battery
2.6.2.6.6.1. basically the same as the voltaic pile
2.6.2.6.6.2. various combinations of materials produce batteries for different uses
2.6.2.6.7. provided steady source of electric current for further use and study
2.6.2.6.7.1. circuits etc.
2.7.1. lightning is electrical discharge and ground
2.7.1.1. exact process of charge separation not well understood
2.7.1.2. Franklin's kite in 17522.7.1.2.1. Richardson killed a year later
2.7.1.3. invented lightning rod to draw charge away from clouds and conduct it away
2.7.2. St. Elmo's fire is a corona discharge from charged objects
2.7.2.1. sheet lightning often occurs within clouds
2.7.3. eels and other fish often produce charge separations
3.1.1. The Prism and the Spectrum
White light is a mixture of colors. When passed through a prism the white light is broken into a spectrum of colors. It is the prismatic effect of tiny drops of rain that produces the spectrum we know as the rainbow. Our modern understanding of color is essentially Newton's. It was this theory that Hooke so ignobly disputed, and which drove Newton away from science.
There are two ways to make color. One is to emit light of a certain color, the other is to remove certain colors from white light. Most of the objects we see either absorb or filter various colors from the natural white light from the sun or from artificial sources.
Although sunlight and artificial light have slightly different spectrums, they all look generally white to us.
All solid objects reflect a certain amount of the light that strikes them. That is how we see them. But generally the color of the light is not the same as the color that strikes it.
Red paper is red because it reflects red light and absorbs the other colors. The color we see is the color that is reflected.
A filter, such as a piece of colored cellophane absorbs all light except the color it transmits. A piece of green cellophane transmits green light and absorbs the rest.
Each substance reflects and transmits a unique combination of colors. That is why there are so many different colors in the world, but it it also why we can use it to identify substances on other planets, in the sun, and in distant stars.
3.2.1. Fraunhofer lines
Fraunhofer Lines. The dark vertical band in the spectrum of the sun represent particular colors of light that are missing. The missing colors correspond to particular wavelengths of light.. You can link here to the Mees Solar Observatory on Maui to see various solar spectra.
3.2.1.1. discovered in spectrum of the sun (Fraunhofer (1787-1826)
3.2.1.2. dark lines in the spectrum indicate selective absorption of certain specific wavelengths
3.2.1.3. interpreted by Bunsen (1811-1899) and Kirchoff (1824-1887)
3.2.1.4. each substance (element or compound) has distinctive fingerprint3.2.1.4.1. absorbed by sun's atmosphere as light passes through it
3.2.2. diffraction grating
The video program demonstrates the use of the diffraction grating to produce a spectrum.
3.2.2.1. uses wave property of light
3.2.2.2. to spread light out over wider area than prism
3.2.2.3. finer resolution of colors
3.2.2.4. more precision in measuring wavelengths3.2.3. continuous vs. discrete spectra
A fluorescent light produces a spectrum which is partially continuous and partially discrete. The discrete spectrum comes from the ultraviolet spectrum of mercury. The visible spectrum is emitted by the phosphor chemical coating on the inside of the bulb.
3.2.3.1. The video program demonstrates continuous and discrete spectra.
3.2.4. emission vs. absorption spectra
3.2.4.1. addition vs. subtraction of specific wavelengths
3.2.4.2. absorbed wavelengths are the same as those emitted by a gas when burned or excited by electricity
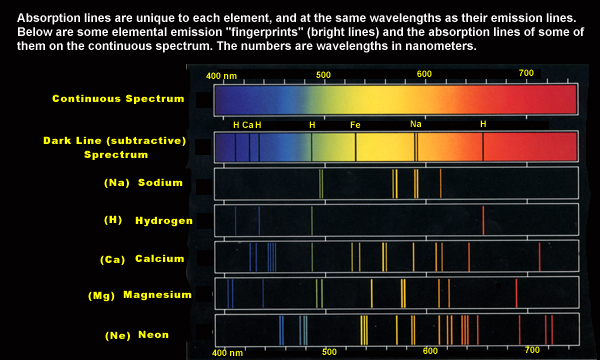
3.3.1. each substance has a unique spectroscopic signature
3.3.2. frequency analysis of light (spectroscopy) allows chemical analysis
3.3.3. in laboratory chemicals
3.3.4. in distant stars, planets, interstellar gas clouds3.3.4.1. emitted, reflected or transmitted light
3.3.5. elements elsewhere in the universe have the same signature as those on Earth
3.3.6. laws of nature are truly universal, the same elsewhere as on Earth
Lines of the Hydrogen Spectrum
4.2.1. in 1885 found simple formula which relates the wavelengths of the various lines
The Balmer Formula
4.2.1.1. Pythagorean or meaningful?
4.2.2. predicted another line corresponding to n = 7 in UV range
4.2.2.1. discovered immediately, others followed for n>7
4.2.3. also predicted that 1/2 could be replaced by 1/1, 1/3, 1/4, etc.
4.2.3.1. lines corresponding to those also found soon thereafter in UV and IR
4.2.4. applied only to the hydrogen spectrum
| The Rydberg Formula. Compare this with the Balmer formula (4.2.1). Here the symbols m and n can be any small integer. |
|
4.4.1. similar to harmonics of a musical tone, but no comparable relationship could be found
4.4.2. cause not understood until details of atomic structure were worked out
Electrostatic attraction is the operating principle of the Xerox machine. A rotating drum becomes electrically charged when bombarded with light. Dark markings, such as writing on a page, do not become charged. The charge is transferred to the paper, passed through a bath of finely ground carbon and plastic known as tone, then baked to solidify and fuse the toner.
Small particles of smoke and other contaminants from industrial fire are attracted to charged metal plates in the smokestack. The remaining gases are discharged into to atmosphere. Although gases are pollutants, they do not cause the haze associated with smoke.
5.3.1. gasoline trucks
Gasoline trucks are grounded to dissipate charges with are separated by friction and stored in the metal tank. The separated charges are immediately lost by contact with the road rather than being concentrated until a spark jumps from the tank to the road. High voltage sparks and gasoline are not a desirable combination.
5.3.2. grain and organic powder storage and movement
Large amounts of grains such as wheat can generate charges by friction as they are poured from one storage bin to another. These organic materials are highly explosive due to the large amount of surface area of the grains, which allows an ample supply of oxygen.